Understanding the periodic table is at the heart of GCSE Chemistry. Whether you’re sitting AQA, Edexcel or OCR, the periodic table is your road‑map to the elements. It shows you how atoms are arranged, why metals behave differently from non‑metals, and how groups and periods reveal patterns in reactivity. Yet each exam board’s insert has subtle differences, and many students overlook simple exam strategies that can save time and marks. This guide unpacks everything you need to know about the GCSE periodic table, explains how it’s structured and used in UK exams, and offers UK‑specific revision tips for 2026.
Jump to Section
Why the periodic table matters in GCSE Chemistry
Chemistry isn’t just about memorising facts. The modern periodic table orders elements by increasing atomic number, the number of protons in an atom into horizontal rows called periods and vertical columns called groups. Elements in the same group share similar properties because they have the same number of electrons in their outer shell.
These patterns help you predict reactions, balance equations and understand why metals lose electrons while non‑metals gain them. Grasping the logic of the table is therefore a powerful exam skill. In 2026, exam boards continue to provide an insert during your exams, but it omits certain rows and uses specific group numbering. Knowing these nuances will help you avoid surprises on test day.
The modern periodic table explained
The periodic table is “periodic” because the pattern of chemical properties repeats at regular intervals. Here are the basics every GCSE candidate should know:
- Periods (rows): Each row corresponds to a new electron shell. As you move left to right across a period the atomic number increases by one and metallic character decreases.
- Groups (columns): Atoms in a group have the same number of outer‑shell electrons, which gives them similar chemical properties. For instance, group 1 (alkali metals) all have one outer electron and react vigorously with water.
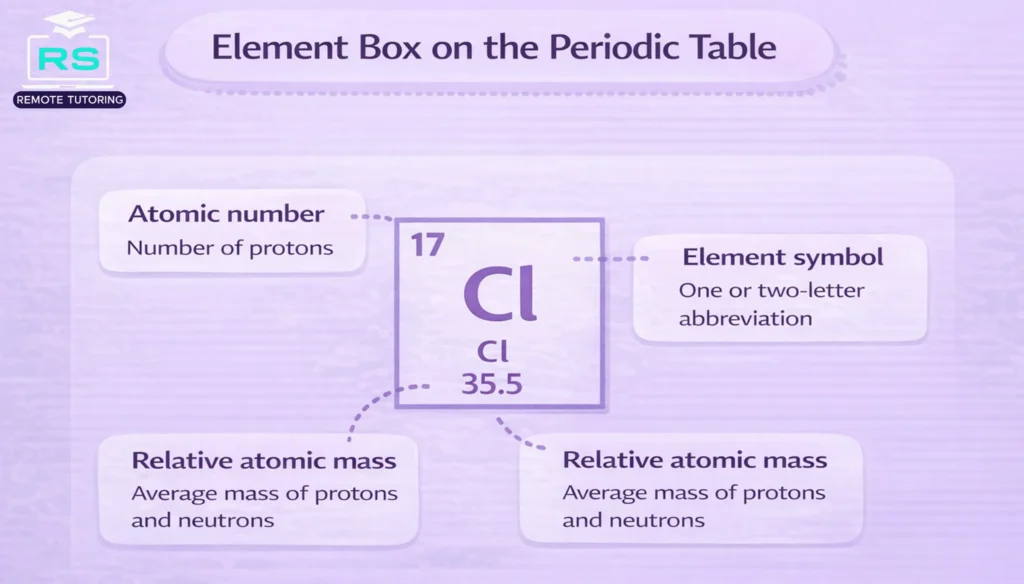
- Atomic number vs relative atomic mass: The smaller number in a periodic table cell is the atomic number, the number of protons. The larger number is the relative atomic mass, roughly the total number of protons and neutrons. In GCSE exams, some atomic masses are not rounded to whole numbers; for example, copper is listed as 63.5 and chlorine as 35.5.
- Lanthanides and actinides: These rows (elements 58 to 71 and 90 to 103) are usually omitted from GCSE tables. You don’t need them for GCSE Chemistry, but you should know they exist.
Metals vs non‑metals
The stepped line on the periodic table separates metals from non‑metals. Metals lie to the left of this line and non‑metals to the right. Group 1 and group 2 are metals; groups 6, 7 and 0 are non‑metals. Metals tend to lose electrons to form positive ions, while non‑metals gain electrons to form negative ions. Most elements are metals, which is why the left of the table is more crowded.
The most important groups
Group 1 – Alkali metals: These reactive metals have one electron in their outer shell and react vigorously with water, oxygen and chlorine. Their reactivity increases down the group: lithium reacts steadily with water while potassium ignites and disappears quickly.
Group 7 – Halogens: These non‑metals exist as diatomic molecules (e.g., Cl₂) and have seven outer electrons. They react with metals to form halide salts and with non‑metals to form covalent compounds. Reactivity decreases down the group because gaining an extra electron becomes harder as the atoms get larger.
Group 0 – Noble gases: These gases have full outer shells (eight electrons, except helium with two) and are unreactive. Their boiling points increase down the group.
How exam boards design the GCSE periodic table

Not all GCSE periodic tables look the same. AQA, Edexcel and OCR use slightly different formats. Creative Chemistry compares them and notes the main differences:
| Feature | AQA | Edexcel | OCR |
| Group numbering | Uses numbers 1 to 0 across the top; recommended IUPAC numbers 1 to 18 are not used | Similar to AQA | OCR uses the IUPAC numbers 1 to 18 for groups |
| Relative atomic mass | Shown below the element symbol; values given to two decimal places; copper and chlorine masses are not rounded | Same as AQA | Same as AQA |
| Atomic number | Shown above the symbol | Same | Same |
| Lanthanides & actinides | La and Ac symbols are shown; lanthanides (58 to 71) and actinides (90 to 103) are omitted | Same | OCR includes the names “lanthanides” and “actinides” for these rows |
| Super‑heavy elements | Elements 112 to 118 (Cn, Fl, Lv) are shown | None shown | Only Cn, Fl and Lv are shown |
These differences don’t change the chemistry, but you should practice with your board’s version so you know where to look for atomic numbers and masses. Some boards provide a printable periodic table or digital insert; make sure to download the correct one for revision.
What AQA’s periodic table insert includes
AQA’s insert for Chemistry GCSE notes that the lanthanides (atomic numbers 58 to 71) and actinides (90 to 103) are omitted, and that the relative atomic masses of copper (Cu) and chlorine (Cl) are not rounded to whole numbers. These exceptions are important because you might need to calculate relative formula mass using 63.5 for Cu and 35.5 for Cl. The insert lists each element with its atomic number and symbol, but does not include the mass number for hydrogen (1) or oxygen (16). Knowing which data are provided will help you plan your answers.
How to use the periodic table during your GCSE exam
You’ll receive a periodic table insert in your exam booklet. Here are some pro tips to make the most of it:
1. Identify metals vs non‑metals quickly: Draw an imaginary zig‑zag line starting at boron (B), down through aluminium (Al) and silicon (Si). Elements to the left are metals; those to the right are non‑metals. Use this to decide whether an element will form positive or negative ions.
2. Use group numbers to predict reactivity: The group number equals the number of outer electrons. Alkali metals (group 1) react more vigorously down the group, while halogens (group 7) get less reactive down the group because larger atoms find it harder to gain an extra electron.
3. Check atomic numbers: If a question asks you about atomic structure, look at the atomic number (above the symbol) to find the number of protons. For example, chlorine has atomic number 17, meaning it has 17 protons and 17 electrons.
4. Use relative atomic masses for calculations: The larger number (below the symbol) is the relative atomic mass; use it to calculate molar masses. Remember that Cu is 63.5 and Cl is 35.5, while most other masses are whole numbers.
5. Remember omissions: Lanthanides and actinides aren’t shown, so don’t worry if you can’t find them. If a question mentions uranium or cerium, you’re likely in the wrong exam!
6. Don’t confuse atomic number with mass number: The smaller number is the atomic number; the larger one is the mass. In GCSE calculations, always use the mass number for Ar/Mr calculations.
Understanding metals and non‑metals for exam questions
Many GCSE questions test your understanding of metallic and non‑metallic properties. Here’s a summary:
Why metals form positive ions
Metals have few electrons in their outer shell and are found on the left side of the periodic table. In reactions, metal atoms give away electrons to form positive ions and attain a stable full shell. This explains why sodium (Na) loses one electron to form Na⁺ and magnesium (Mg) loses two to form Mg²⁺. Losing electrons is easier down a group because the outer electron is further from the nucleus and more shielded by inner electrons; hence reactivity increases down group 1.
Why non‑metals form negative ions
Non‑metals are on the right and have more electrons in their outer shells. They gain electrons to complete their outer shell, forming negative ions. For instance, chlorine (Cl) gains one electron to become Cl⁻. Going down group 7, halogen atoms get larger and the attractive force on the incoming electron decreases, so reactivity decreases. That’s why chlorine is more reactive than bromine and iodine.
Why noble gases are inert
Group 0 elements have full outer shells, so they are stable and don’t easily gain or lose electrons. They are used in lighting and signage because they do not react.
Working with exam board differences: AQA vs Edexcel vs OCR
Although all boards test the same core concepts, their periodic table inserts and question styles differ slightly. Here’s how to navigate them:
AQA: The group numbers are 1 to 0, and relative atomic masses are given to two decimal places. Lanthanides and actinides are omitted. Because copper and chlorine masses aren’t rounded, always use 63.5 and 35.5 in your calculations.
Edexcel: Similar to AQA for Chemistry, though some Edexcel papers (especially International GCSE) may provide a different arrangement or omit super‑heavy elements. Practice with the Edexcel insert to recognise the layout. Past papers often emphasise calculations and practical interpretation.
OCR: Uses IUPAC group numbers 1 to 18. Super‑heavy elements beyond 111 may not be shown. OCR questions often involve applying periodic trends to unfamiliar elements; understanding group and period patterns is essential.
Which periodic table should you revise from?
Always use the insert provided by your exam board. You can download AQA’s periodic table insert or printable versions from their website. For Edexcel and OCR, check the exam support page. Printing a copy to stick above your desk or on your fridge can reinforce learning.
Pro tip: practise interpreting the table
When you revise, don’t just memorise the layout. Practise using the table to answer questions:
- Identify element positions quickly (period and group).
- Predict whether an element is metal or non‑metal.
- Use atomic numbers to deduce electron configurations.
- Use relative atomic masses to calculate Mr for compounds.
Seasonal and UK‑specific exam advice
In the UK, GCSE Chemistry exams are usually sat in late May and June. The 2026 sessions follow the same pattern, with paper 1 typically in mid‑May and paper 2 in early June. Revision should start early in the academic year. Winter evenings are long in the UK, so use the extra darkness to create a study schedule that avoids burnout. As daylight increases in spring, allocate more time to past papers and timed questions to simulate exam conditions. Don’t forget to check your exam timetable, AQA, Edexcel and OCR publish provisional dates in January.
If you live in the north of the UK, keep in mind that exam mornings can be chilly and damp in May. Plan your journey to the exam centre with traffic and weather in mind. Arrive early so you have time to relax and read the instructions on your periodic table insert.
Frequently Asked Questions
What grade is 75% in Chemistry GCSE?
Exam boards set grade boundaries after papers are marked. In recent years, a score around 75% has typically fallen into the grade 7 or 8 range for higher‑tier Chemistry. For example, in 2025 AQA and Edexcel maths papers required around 75–80% for a grade 8. Chemistry boundaries are similar but may vary each year. Always check the published grade boundaries after results day to see how your score translates to a grade.
Is element 119 possible?
Scientists have synthesised elements up to atomic number 118 (oganesson). Element 119, temporarily named ununennium, has not yet been officially discovered or verified. Researchers continue to attempt to create it by colliding lighter nuclei, but its existence would be extremely short‑lived because super‑heavy elements decay quickly. It is not part of any GCSE curriculum.
How hard is it to get a 9 in GCSE Chemistry?
Achieving a grade 9 requires a deep understanding of the specification and excellent exam technique. Only the top few percent of students receive a 9. Typically, you need to score above 85–90% overall, depending on the paper difficulty. Focus on mastering the content, practising past papers and explaining concepts clearly.
What do I need to know about the periodic table in GCSE?
You should know that elements are arranged in order of increasing atomic number and grouped by similar properties. Understand that metals are on the left and form positive ions, while non‑metals are on the right and form negative ions. Learn the trends in group 1, group 7 and group 0, and be able to use the periodic table to determine atomic numbers, relative atomic masses, electron configurations and element positions. Also be aware that your exam insert omits lanthanides and actinides.
Is 80% a 9 in GCSE?
A score of 80% often corresponds to a grade 8 on higher‑tier papers, though it can sometimes be enough for a grade 9 if the paper was particularly challenging. Grade boundaries vary each year, so check the official boundaries published on results day to know exactly.
What grade is 63% at GCSE?
A mark of 63% usually falls around grade 6 or 7 on higher‑tier papers. For example, 63% is often between the grade 6 and grade 7 boundaries on AQA and Edexcel science papers. Boundaries change yearly, so consult the official grade boundary tables once released.
Is a grade 9 top 5%?
Approximately the top 5 to 7% of candidates achieve a grade 9. Ofqual designs the 9‑1 grading scale so that grade 9 recognises exceptional performance comparable to the top half of the old A* grade. The exact percentage of students achieving a 9 can vary slightly between subjects and years.
What grade is 60% in the UK?
In GCSEs, a score of around 60% generally results in a grade 5 or 6, depending on the subject and paper difficulty. Grade 5 is regarded as a “strong pass,” while grade 4 is a “standard pass.” Always consult official grade boundaries once they are published for your subject.
Conclusion
Mastering the periodic table is about understanding patterns, not memorising every detail. If you want personalised help to boost your Chemistry grade, our expert tutors in the UK can guide you through tricky concepts, exam techniques and revision planning. Contact us today to book a free consultation and take the next step toward your GCSE success.

Raja specializes in Physics and Maths, with over 5 years of experience. He offers KS2, KS3, and GCSE Science and Maths lessons. He graduated from one of the top universities in the UK.








